Surfaces interaction data, allergy cross-references, and contraindication information — powered by MIMS.
Echo-Health surfaces medication safety information during consultations using MIMS pharmaceutical database integration. When a clinician discusses or reviews medications, the system highlights relevant interaction data, allergy cross-references, and contraindication information — presented for clinician review within the consultation workflow.
When a mentioned medication conflicts with a documented allergy, Echo-Health surfaces the alert with source information. Allergy data is drawn from the patient record via FHIR, and the cross-reference is presented alongside the relevant consultation context for clinician review.
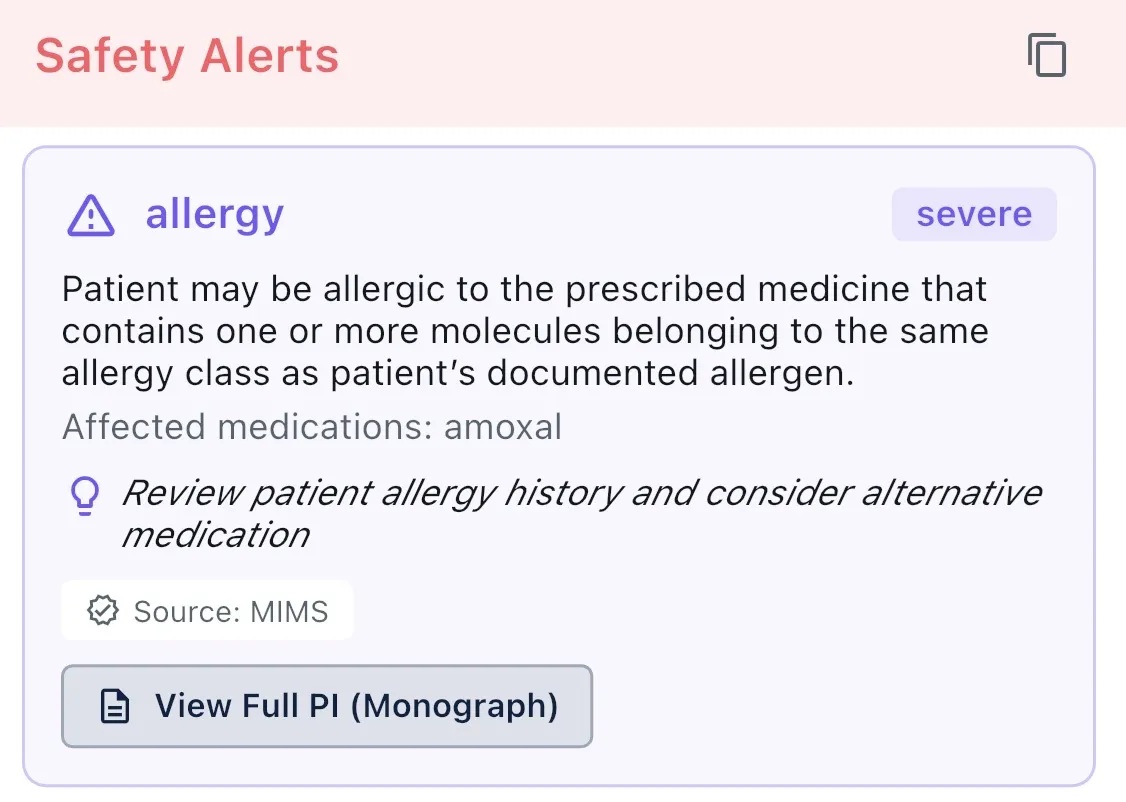
Surfaces interaction data between current medications and new prescriptions — including severity and clinical significance. Information is sourced from MIMS pharmaceutical databases and presented for clinician review during the consultation.

Clinicians can access full MIMS product information directly within the consultation workflow. Detailed pharmaceutical data — including indications, dosage, contraindications, and interaction profiles — is available without leaving the consultation screen.
Echo-Health's evidence layer is constrained by architectural guardrails that enforce clinical governance at the system level — not through policy alone.
Every clinical statement must include a PMID and DOI where available. If no evidence is found, the system says so clearly — it never infers or extrapolates.
“Should”, “recommended”, “first-line”, “start with”, “dose”, “titrate” — all prohibited from evidence output at the system architecture level.
The evidence layer retrieves and summarises published literature only. It does not provide clinical advice, evaluation, or treatment recommendations.
Full audit trail for every evidence query: search terms, article counts, model versions, timestamps, and complete reference lists.
Echo-Health surfaces four categories of medication safety information during the consultation — powered by integrated pharmaceutical databases. All information is presented for clinician review.
Echo-Health's evidence and safety layers are designed for organisations with clinical governance requirements. Architectural constraints — not just policies — ensure the system cannot cross into clinical recommendations. Contact us for a clinical governance briefing.
All safety information is presented for clinician review. Echo-Health does not make clinical decisions. Medication safety information is sourced from MIMS pharmaceutical databases and is intended to support — not replace — clinical judgement.